0.0055% serious side effects following 17.7 million doses in Canada
Reported side effects following COVID-19 vaccination in Canada
[Government of Canada.] There have been a total of 5,488 reports (31.0 reports per 100,000 doses administered) up to and including May 14, 2021. Of the 5,488 reports, 977 were considered serious (5.5 reports per 100,000 doses administered [0.0055%]). While the number of doses administered have increased over time, the rate of serious reports has remained low.
Effectiveness of COVID-19 vaccines against the B.1.617.2 variant
[Indian.] Data on all symptomatic sequenced cases of COVID-19 in England was used to estimate the proportion of cases with B.1.617.2 [Indian] compared to the predominant strain (B.1.1.7 [UK]) by vaccination status. Effectiveness was notably lower after 1 dose of vaccine with B.1.617.2 cases 33.5% (95%CI: 20.6 to 44.3) compared to B.1.1.7 cases 51.1% (95%CI: 47.3 to 54.7) with similar results for both vaccines. With BNT162b2 2 dose effectiveness reduced from 93.4% (95%CI: 90.4 to 95.5) with B.1.1.7 to 87.9% (95%CI: 78.2 to 93.2) with B.1.617.2. With ChAdOx1 2 dose effectiveness reduced from 66.1% (95% CI: 54.0 to 75.0) with B.1.1.7 to 59.8% (95%CI: 28.9 to 77.3) with B.1.617.2. Sequenced cases detected after 1 or 2 doses of vaccination had higher odds of infection with B.1.617.2 compared to unvaccinated cases (OR 1.40; 95%CI: 1.13-1.75).
Diverse Functional Autoantibodies in Patients with COVID-19
We found that COVID-19 patients exhibit dramatic increases in autoantibody reactivities compared to uninfected controls, with a high prevalence of autoantibodies against immunomodulatory proteins including cytokines, chemokines, complement components, and cell surface proteins. We established that these autoantibodies perturb immune function and impair virological control by inhibiting immunoreceptor signaling and by altering peripheral immune cell composition, and found that murine surrogates of these autoantibodies exacerbate disease severity in a mouse model of SARS-CoV-2 infection.
Ohio sees surge in vaccination shots after offering $5 million lottery
[Media report.] Health officials in Ohio have reported a surge […] a week after Ohio announced the $5 million "Vax-a-Million" lottery. Based on preliminary data, the department said the recent period showed a 53% week-to-week increase (May 13 to 18) compared to the time period before the announcement, where 74,000 people received their first dose (May 6 to 11). [See also NY 'Vax & Scratch' Program.]
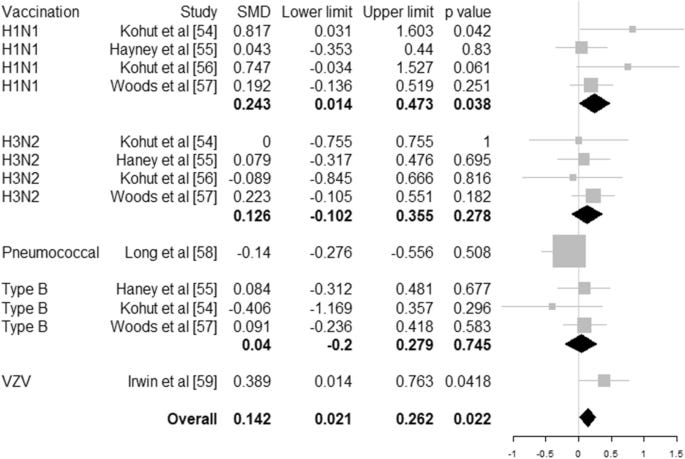
Effects of Regular Physical Activity on the Immune System, Vaccination and Risk of Community-Acquired Infectious Disease in the General Population: Systematic Review and Meta-Analysis
We searched seven databases up to April 2020. Higher level of habitual physical activity is associated with a 31% risk reduction (hazard ratio 0.69, 95% CI 0.61–0.78, 6 studies, N = 557,487 individuals) of community-acquired infectious disease and 37% risk reduction (hazard ratio 0.64, 95% CI 0.59–0.70, 4 studies, N = 422,813 individuals) of infectious disease mortality. Physical activity interventions resulted in increased CD4 cell counts (32 cells/µL, 95% CI 7–56 cells/µL, 24 studies, N = 1112 individuals) and salivary immunoglobulin IgA concentration (standardised mean difference 0.756, 95% CI 0.146–1.365, 7 studies, N = 435 individuals) and decreased neutrophil counts (704 cells/µL, 95% CI 68–1340, 6 studies, N = 704 individuals) compared to controls. Antibody concentration after vaccination is higher with an adjunct physical activity programme (standardised mean difference 0.142, 95% CI 0.021–0.262, 6 studies, N = 497 individuals). Regular, moderate to vigorous physical activity is associated with reduced risk of community-acquired infectious diseases and infectious disease mortality, enhances the first line of defence of the immune system, and increases the potency of vaccination.