2/19—Delayed vaccine second dose: Pfizer 92.6% effective; Moderna 92.1%
Delayed Second Dose versus Standard Regimen for Covid-19 Vaccination
You chair the Governor’s task force on rollout of the Covid-19 vaccine. Which one of the following approaches would you take?
Recommend Delaying the Second Dose
Robert M. Wachter, M.D.
The clinical trials of the Pfizer–BioNTech and Moderna vaccines involved two injections given 3 to 4 weeks apart. Both vaccines had approximately 95% efficacy after the second dose — an impressive finding. In both trials, the cases in the placebo and active vaccine groups began to diverge about 10 days after the first dose, with growing vaccine efficacy over time. By the day of the injection of the second dose, the efficacy of the first dose was somewhere in the range of 80 to 90%. One model shows that the expected number of Covid-19 cases would be significantly lower if more people were given a first dose, even if it came at the cost of deferring the second doses. [Also,] we have recently seen the emergence of several viral variants, with one (B.1.1.7, often referred to as the U.K. variant) that is approximately 50% more infectious than the native coronavirus.
Recommend Following the Standard Regimen
Nicole Lurie, M.D., M.S.P.H.
Although the immune response to the first dose is unlikely to degrade quickly, it is incomplete, and there are no data to inform how long a second dose could be delayed without compromising effectiveness. Some models have suggested that using a less effective vaccine or delaying a second dose to provide first doses to more people will end the pandemic sooner. However, these models do not account for the potential degradation of the immune response or for spillover effects of such decisions on vaccine acceptance. Many people are skeptical of vaccines, fearing that the speed of development has necessitated cutting corners and that political pressure has influenced vaccine recommendations. Suddenly changing dosing recommendations puts public confidence at serious risk and will impede willingness to be vaccinated at all. Cases of Covid-19 have already occurred in vaccine recipients, as was seen in the phase 3 trials, which will raise questions about the delayed-second-dose strategy and erode trust in the vaccine rollout. The appearance of SARS-CoV-2 variants implies that the virus is under evolutionary pressure. Some have postulated — although this is speculative — that subinhibitory levels of antibody response before a second dose, if widespread, could contribute to selection of antigenic variants that could escape current vaccines.
Correspondence: Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine [Pfizer]
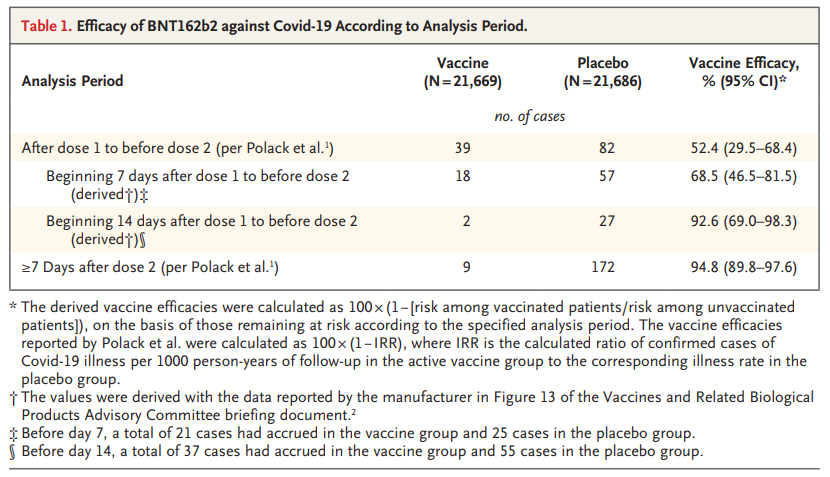
We used documents submitted to the Food and Drug Administration to derive the vaccine efficacy beginning from 2 weeks after the first dose to before the second dose (Table 1). Even before the second dose, BNT162b2 was highly efficacious, with a vaccine efficacy of 92.6%, a finding similar to the first-dose efficacy of 92.1% reported for the mRNA-1273 vaccine (Moderna). With such a highly protective first dose, the benefits derived from a scarce supply of vaccine could be maximized by deferring second doses until all priority group members are offered at least one dose.
Authors reply: We would like to emphasize that alternative dosing regimens of BNT162b2 have not been evaluated. The decision to implement alternative dosing regimens resides with health authorities.
Correspondence: Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine [Pfizer]
Polack et al. may have erroneously concluded that the differences in the absolute numbers of severe Covid-19 cases between the vaccine group and the placebo group provide preliminary evidence of protection against the development of severe Covid-19 illness. The percentage of Covid-19–positive patients in whom severe illness developed was 5.6% (9 of 162 patients) in the placebo group and 12.5% (1 of 8 patients) in the vaccine group — a difference of 6.9 percentage points (95% confidence interval [CI], 6.4 to 7.6) (P<0.001 by the chi-square test of proportions).
Authors reply: We would like to clarify that it is not appropriate to use the proportion of Covid-19–positive patients in whom severe disease developed to assess vaccine protection against severe Covid-19. Protection against severe illness is an integrated effect of reducing the chance that any Covid-19 symptom will develop and reducing the risk that severe symptoms will develop after infection. The calculation provided considers only the second effect, and the estimate for the vaccine group is very imprecise owing to the small sample size (only 8 cases in this group). After the first dose, vaccine efficacy against the development of severe Covid-19, calculated as 100×(1–IRR), where IRR is the ratio of confirmed cases of severe Covid-19 illness per 1000 person-years of follow-up for the active vaccine group to the corresponding illness rate in the placebo group, was 88.9% (95% CI, 20.1 to 99.7).
Missed an update? View past issues.