3/10—Vaccinating previously infected individuals
Antibody response to first BNT162b2 dose in previously SARS-CoV-2-infected individuals [Pfizer]
We undertook a nested case-control analysis of 51 health-care workers in London. All participants received their first dose of the BNT162b2 mRNA COVID-19 vaccine (Pfizer-BioNTech, Mainz, Germany), and were tested 19–29 days later (median 22 days, IQR 2). Among previously uninfected, seronegative individuals, anti-S titres after one vaccine dose were comparable to peak anti-S titres in individuals with a previous natural infection who had not yet been vaccinated. Among those with a previous SARS-CoV-2 infection, vaccination increased anti-S titres more than 140-fold from peak pre-vaccine levels (figure). This increase appears to be at least one order of magnitude greater than reported after a conventional prime-boost vaccine strategy in previously uninfected individuals. These serological data suggest that for individuals receiving the BNT162b2 mRNA vaccine, a potential approach is to include serology testing at or before the time of first vaccination to prioritise use of booster doses for individuals with no previous infection.
Effect of previous SARS-CoV-2 infection on humoral and T-cell responses to single-dose BNT162b2 vaccine [Pfizer]
We assessed immune responses to spike protein (S) post-vaccination. Anti-S titres were significantly higher in individuals with previous natural infection than in infection-naive individuals (median 16353 arbitrary units [AU] per mL [IQR 4741–28 581] vs 615·1 AU/mL (286·4–1491), p<0·0001; figure A). The five participants with previous natural infection yet negative serology at baseline developed post-vaccination anti-S titres that were intermediate between the infection-naive and previously infected groups (figure A). We show that individuals with previous SARS-CoV-2 infection generate strong humoral and cellular responses to one dose of BNT162b2 vaccine, with evidence of high titres of in-vitro live virus neutralisation. In contrast, most individuals who are infection-naive generate both weak T-cell responses and low titres of neutralising antibodies.
Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials [AstraZeneca]
We present data from three single-blind randomised controlled trials and one double-blind study in South Africa. Between April 23 and Dec 6, 2020, 24 422 participants were recruited and vaccinated across the four studies. Overall vaccine efficacy more than 14 days after the second dose was 66·7% (95% CI 57·4–74·0), with 84 (1·0%) cases in the 8597 participants in the ChAdOx1 nCoV-19 group and 248 (2·9%) in the 8581 participants in the control group. There were no hospital admissions for COVID-19 in the ChAdOx1 nCoV-19 group after the initial 21-day exclusion period, and 15 in the control group. 108 (0·9%) of 12 282 participants in the ChAdOx1 nCoV-19 group and 127 (1·1%) of 11 962 participants in the control group had serious adverse events. There were seven deaths considered unrelated to vaccination (two in the ChAdOx1 nCov-19 group and five in the control group), including one COVID-19-related death in one participant in the control group. The results of this primary analysis of two doses of ChAdOx1 nCoV-19 were consistent with those seen in the interim analysis of the trials and confirm that the vaccine is efficacious, with results varying by dose interval in exploratory analyses. A 3-month dose interval might have advantages over a programme with a short dose interval for roll-out of a pandemic vaccine to protect the largest number of individuals in the population as early as possible when supplies are scarce, while also improving protection after receiving a second dose.
Effectiveness of BNT162b2 mRNA Vaccine Against Infection and COVID-19 Vaccine Coverage in Healthcare Workers in England, Multicentre Prospective Cohort Study (the SIREN Study) [Pfizer]
[Preprint.] A single dose of BNT162b2 vaccine demonstrated vaccine effectiveness of 72% (95% CI 58-86) 21 days after first dose and 86% (95% CI 76-97) seven days after two doses in the antibody negative cohort. Our study demonstrates that the BNT162b2 vaccine effectively prevents both symptomatic and asymptomatic infection in working age adults; this cohort was vaccinated when the dominant variant in circulation was B1.1.7 and demonstrates effectiveness against this variant.
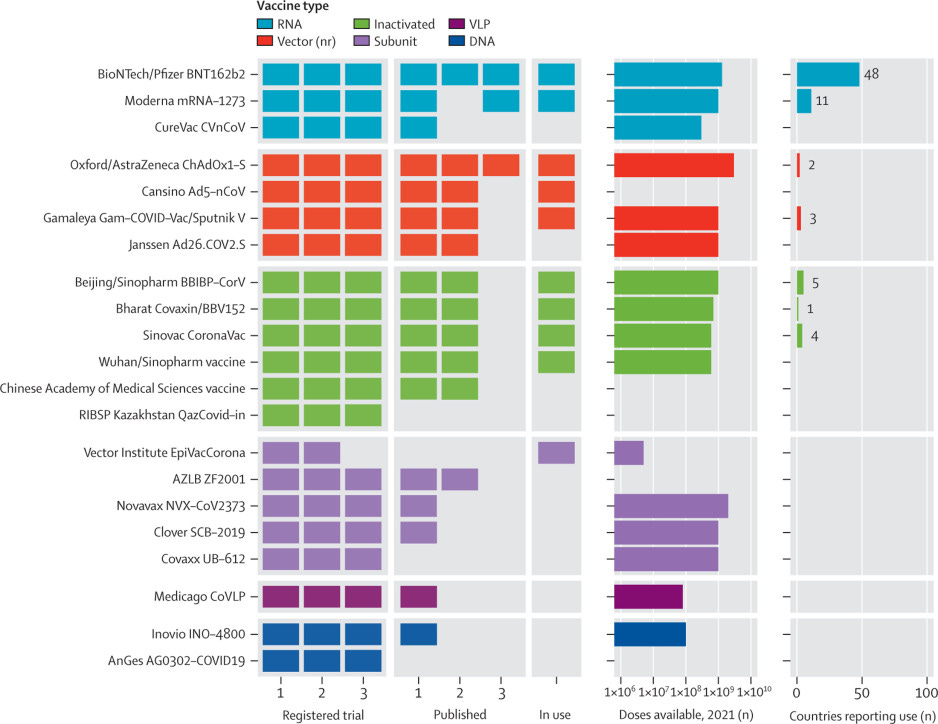
An interactive website tracking COVID-19 vaccine development
We developed an online, interactive vaccine tracker hosted by the Vaccine Centre (VaC) at the London School of Hygiene and Tropical Medicine (LSHTM; London, UK). The tracker includes a clinical trials database that captures specifications such as locations, dates, size, study design, masking, randomisation, and recruitment status of registered COVID-19 vaccine trials in humans. Newly registered or updated protocols are identified weekly. [Visit website.]