4/16—Johnson & Johnson vaccine and blood clots
Joint CDC and FDA Statement on Johnson & Johnson COVID-19 Vaccine
[Joint statement.] As of April 12, more than 6.8 million doses of the Johnson & Johnson (Janssen) vaccine have been administered in the U.S. CDC and FDA are reviewing data involving six reported U.S. cases of a rare and severe type of blood clot in individuals after receiving the J&J vaccine. In these cases, a type of blood clot called cerebral venous sinus thrombosis (CVST) was seen in combination with low levels of blood platelets (thrombocytopenia). All six cases occurred among women between the ages of 18 and 48, and symptoms occurred 6 to 13 days after vaccination. Right now, these adverse events appear to be extremely rare.
Johnson & Johnson Statement on COVID-19 Vaccine
[Press release.] We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine. In addition, we have been reviewing these cases with European health authorities. We have made the decision to proactively delay the rollout of our vaccine in Europe.
EU reviews J&J COVID-19 vaccine for blood clots, expands AstraZeneca probe
[Media report.] Four serious cases of rare blood clots with low platelets, one of which was fatal, have been reported after inoculation with J&J's vaccine from its Janssen unit, the European Medicines Agency said.
Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination [AstraZeneca]
We assessed the clinical and laboratory features of 11 patients in Germany and Austria in whom thrombosis or thrombocytopenia had developed after vaccination with ChAdOx1 nCov-19. Of the 11 original patients, 9 were women, with a median age of 36 years (range, 22 to 49). Beginning 5 to 16 days after vaccination, the patients presented with one or more thrombotic events, with the exception of 1 patient, who presented with fatal intracranial hemorrhage. Of the patients with one or more thrombotic events, 9 had cerebral venous thrombosis, 3 had splanchnic-vein thrombosis, 3 had pulmonary embolism, and 4 had other thromboses; of these patients, 6 died. Vaccination with ChAdOx1 nCov-19 can result in the rare development of immune thrombotic thrombocytopenia mediated by platelet-activating antibodies against PF4, which clinically mimics autoimmune heparin-induced thrombocytopenia.
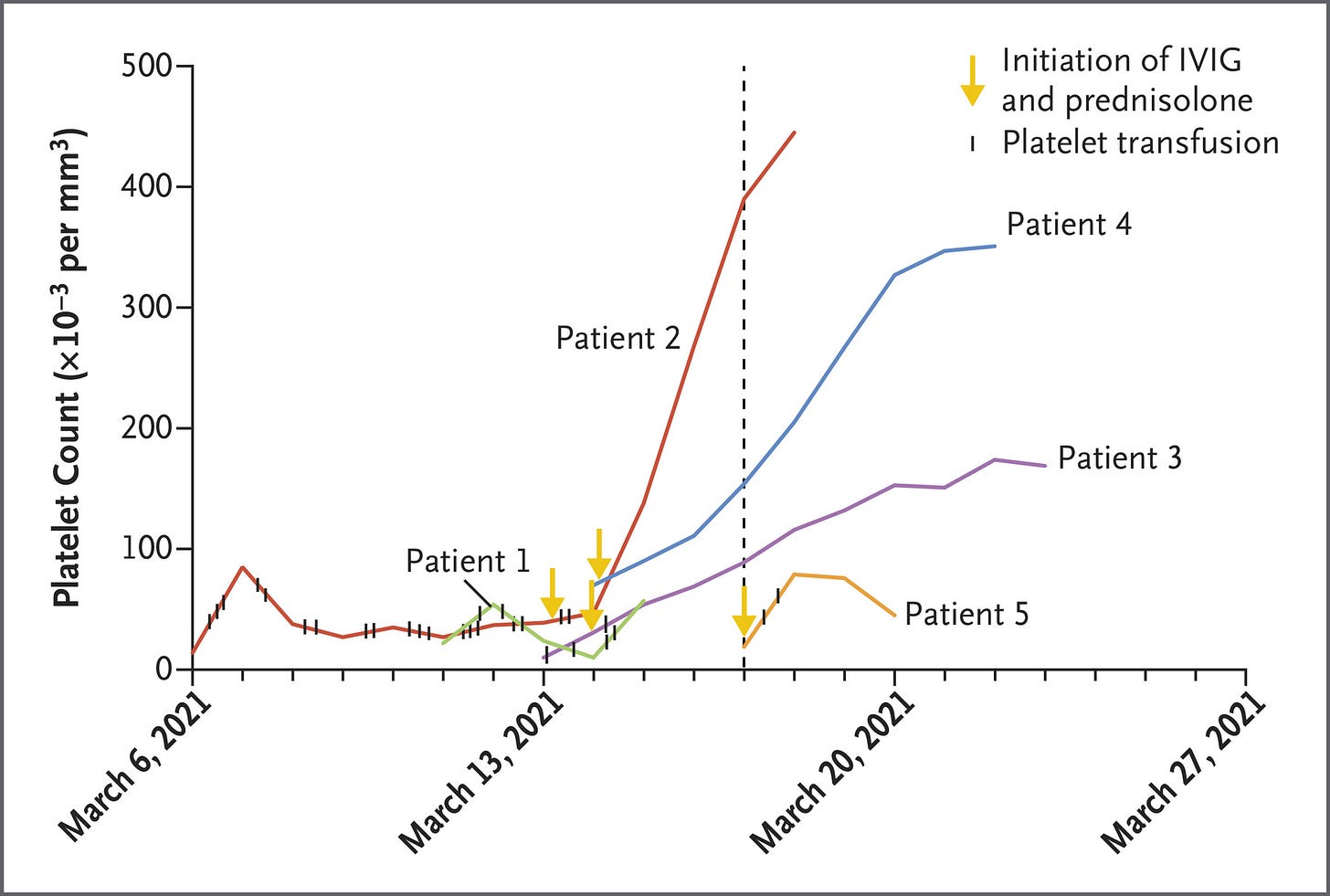
Thrombosis and Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination [AstraZeneca] We report findings in five patients who presented with venous thrombosis and thrombocytopenia 7 to 10 days after receiving the first dose of the ChAdOx1 nCoV-19 adenoviral vector vaccine against Covid-19. The patients were health care workers who were 32 to 54 years of age. Four of the patients had severe cerebral venous thrombosis with intracranial hemorrhage, and the outcome was fatal in three. Although rare, [this] is a new phenomenon with devastating effects for otherwise healthy young adults and requires a thorough risk–benefit analysis.