Booster shots: what is the evidence?
What’s the evidence for COVID-19 booster shots?
As of Aug. 15, Israel reported 514 hospitalizations for severe or critical COVID-19 — up 31% from four days earlier — and nearly three in five of those patients were fully vaccinated. That country is now offering boosters to people over 50. One study of residents at nearly 15 000 long-term care facilities found that the protection against infection offered by the Pfizer and Moderna vaccines dropped from 74% in the spring to 53% this summer. A New York state study that also included people who received the Johnson & Johnson vaccine likewise reported a drop in protection against lab-confirmed infection from 92% in early May to 80% in late July. However, effectiveness against COVID-19 requiring hospitalization held steady at about 95%. Meanwhile, a preprint Mayo Clinic analysis of more than 80 000 Americans found the Pfizer vaccine was only 42% effective against SARS-CoV-2 infection in July, while Moderna’s was 76% effective. Even so, protection against hospitalization remained high at 75% and 81%, respectively. These findings indicate that COVID-19 vaccines are still doing an excellent job keeping fully immunized people out of hospital, even if larger numbers are testing positive for SARS-CoV-2 infection (many of whom may have no symptoms).
Considerations in boosting COVID-19 vaccine immune responses
[Via CIDRAP News.] Observational studies consistently show that the vaccines are still highly effective against severe disease, including severe disease from all variants, with efficacy averaging 80% against any disease. Vaccine efficacy is greater against severe disease than against mild illness. Even in highly vaccinated populations, those who are unvaccinated are still the main drivers of transmission and are at most risk for severe disease. […] Taken together, the studies don't show credible evidence of declining protection against severe disease—vaccination's main goal.
BNT162b2 vaccine booster dose protection: A nationwide study from Israel
[Preprint.] On July 30, 2021, a third (booster) dose of the Pfizer BNT162b2 vaccine was
approved in Israel for individuals 60 years or older who had been fully vaccinated (i.e., received two doses) at least five months previously. Here, we estimate the reduction in relative risk for confirmed infection and severe COVID-19 provided by the booster dose. 1,144,690 individuals aged 60 y and older who were eligible for a booster dose were followed between July 30 and August 22, 2021. Twelve days or more after the booster dose we found an 11.4-fold (95% CI: [10.0, 12.9]) decrease in the relative risk of confirmed infection, and a >10-fold decrease in the relative risk of severe illness. Under a conservative sensitivity analysis, we find ≈5-fold protection against confirmed infection.
Immunogenicity and safety of a third dose, and immune persistence of CoronaVac vaccine in healthy adults aged 18-59 years: interim results from a double-blind, randomized, placebo-controlled phase 2 clinical trial
[Preprint.] Overall, 540 participants received a third dose. In the 3 μg group, neutralizing antibody titers induced by the first two doses declined after 6-8 months to below the seropositive cutoff. When a third dose was given 6-8 months after a second dose, geometric mean titers (GMTs) assessed 14 days later increased to 137.9 [95%CI 99.9-190.4] for Schedule 2, and 143.1 [95%CI 110.8-184.7] for Schedule 4. A third dose of CoronaVac administered 6 or more months after a second dose effectively recalled specific immune response to SARS-CoV-2, resulting in a remarkable increase in antibody levels, and indicating that a two-dose schedule generates good immune memory. Optimizing the timing of a booster dose should take into account immunogenicity, vaccine efficacy/effectiveness, local epidemic situation, infection risk, and vaccine supply.
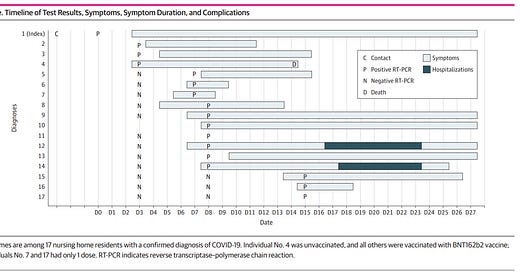
Investigation of an Outbreak of COVID-19 in a French Nursing Home With Most Residents Vaccinated
The study reports an outbreak that occurred in a 77-bed NH located in Biscarrosse, France, in which a vaccine campaign was conducted using the BNT162b2 vaccine. Among 74 residents (16 men [22.2%]; mean [SD] age, 87.8 [7.5] years), 72 individuals (97.3%) were vaccinated, including 70 residents who were fully vaccinated and 2 residents who were partially vaccinated. In total, 17 residents (23.0%) were diagnosed with COVID-19 (5 men [29.4%] and 12 women [70.6%]; mean [SD] age, 87.6 [8.9] years). The individuals diagnosed with COVID-19 included 1 unvaccinated resident, 2 partially vaccinated residents, and 14 fully vaccinated residents. Among individuals with diagnoses, 8 residents developed severe disease, 2 were hospitalized, and 1 individual (the unvaccinated resident) died (Figure). Among 102 health care professionals, 34 individuals were vaccinated (33.3%), 68 individuals were unvaccinated (66.7%), and 12 individuals were diagnosed with COVID-19 (11.8%). Of these, 5 individuals became symptomatic and none had severe disease. Among health care professionals who were unvaccinated, 9 individuals (13.2%) were diagnosed with COVID-19, and among health care professionals who were previously vaccinated, 3 individuals (8.8%) were diagnosed (including 1 individual fully vaccinated with the BNT162b2 vaccine and 2 individuals partially vaccinated with the ChAdOx1 nCoV-19 vaccine). The SARS-CoV-2 variant B.1.1.7 was identified among all residents and health care professionals who were diagnosed with COVID-19. Our study’s findings suggest that SARS-CoV-2 vaccination may not be sufficient as the sole means to prevent COVID-19 among NH residents and that other prevention measures should not be abandoned yet in these settings.