Delaying the second dose to 12 weeks may 3.5x antibody response in people aged 80+
Extended interval BNT162b2 vaccination enhances peak antibody generation in older people
[Pfizer. Preprint.] Setting: Primary care networks, Birmingham, UK. December 2020 to April 2021. Participants: 175 people aged over 80 years of age. All donors received the BNT162b2 Pfizer/BioNTech vaccination and were vaccinated with either a standard 3 week interval between doses or an extended interval schedule. Main outcome measures: Peak quantitative spike-specific antibody and cellular immune responses. Results: In donors without evidence of previous infection the peak antibody response was 3.5-fold higher in donors who had undergone delayed interval vaccination. Cellular immune responses were 3.6-fold lower. Conclusion: Peak antibody responses after the second BNT162b2 vaccine are markedly enhanced in older people when this is delayed to 12 weeks although cellular responses are lower. Extended interval vaccination may therefore offer the potential to enhance and extend humoral immunity. Further follow up is now required to assess long term immunity and clinical protection.
Waning antibodies to SARS-CoV-2 – Don't panic
It is widely accepted that neutralizing antibodies will likely play an important part in the prevention of reinfection. However, there are several other compartments of the immune system that likely contribute to the prevention of clinical disease, especially in its more severe forms. Interestingly, the efficacy trials for mRNA vaccines have shown that protection from symptomatic COVID-19 disease starts around 10-12 days after administration of the first dose, i.e. at a time when there are very few neutralizing antibodies present indicating that the induction of T-cell responses likely play a role in prevention of symptomatic COVID-19. In line with this observation several studies found a strong and lasting induction of memory B and T-cell in SARS-CoV-2 infected patients at 6 months post onset even in asymptomatic individuals.
SARS-CoV-2 vaccines: Lights and shadows
So far, vaccines to prevent SARS-CoV-2 infection appear as promising approach for curbing the pandemic. [But] whereas Phase III vaccine trials generally excluded participants with previous immunization, vaccination of huge populations in the real life will inevitably include individuals with preexisting immunity. This might lead to excessively enhanced inflammatory and thrombotic reactions in occasional subjects. Further research is urgently needed in this area.
Investigate the origins of COVID-19
As scientists with relevant expertise, we agree with the WHO director-general, the United States and 13 other countries, and the European Union that greater clarity about the origins of this pandemic is necessary and feasible to achieve. We must take hypotheses about both natural and laboratory spillovers seriously until we have sufficient data. A proper investigation should be transparent, objective, data-driven, inclusive of broad expertise, subject to independent oversight, and responsibly managed to minimize the impact of conflicts of interest.
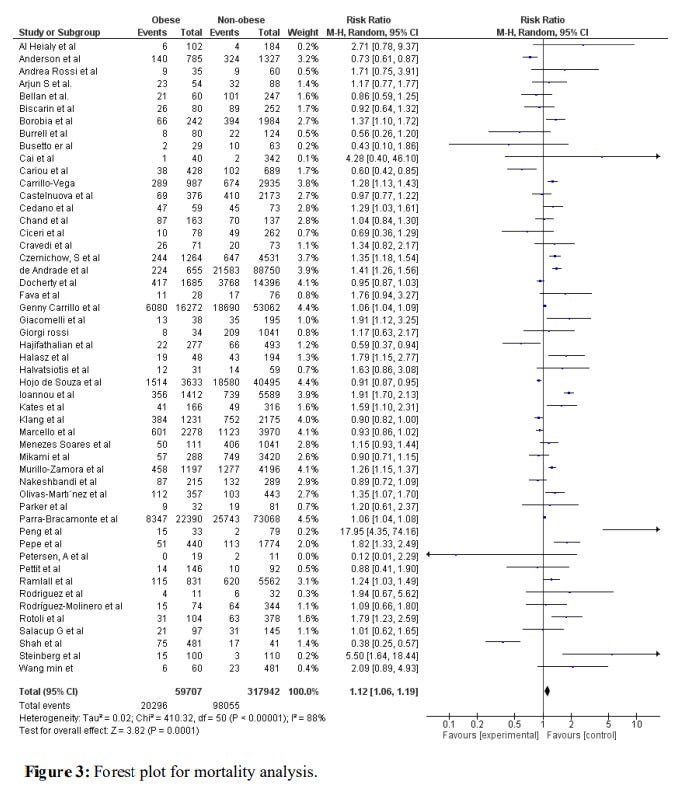
Association of Obesity with COVID-19 Severity and Mortality: A Systemic Review and Meta-Regression
[Preprint.] A total of 576,784 patients from 100 studies were included in this meta-analysis. Being obese was associated with increased risk of severe disease (RR=1.46, 95% CI 1.34-1.60, p<0.001, I2 = 92 %). Similarly, high mortality was observed in obese patients with COVID-19 disease (RR=1.12, 95% CI 1.06-1.19, p<0.001, I2 = 88%). For mortality outcome, covariate of female gender, proportion of pulmonary disease, diabetes, hypertension, and cardiovascular diseases were significant, these covariates collectively explained R2=53% of the between-study variability for mortality.