Effectiveness of vaccines against variants
Effectiveness of the BNT162b2 Covid-19 Vaccine against the B.1.1.7 and B.1.351 Variants
[Pfizer.] Vaccination scale-up occurred as Qatar was undergoing its second and third waves of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, which were triggered by expansion of the B.1.1.7 variant (starting in mid-January 2021) and the B.1.351 variant (starting in mid-February 2021). The estimated effectiveness of the vaccine against any documented infection with the B.1.1.7 variant was 89.5% (95% confidence interval [CI], 85.9 to 92.3) at 14 or more days after the second dose (Table 1 and Table S2). The effectiveness against any documented infection with the B.1.351 variant was 75.0% (95% CI, 70.5 to 78.9). Vaccine effectiveness against severe, critical, or fatal disease due to infection with any SARS-CoV-2 (with the B.1.1.7 and B.1.351 variants being predominant within Qatar) was very high, at 97.4% (95% CI, 92.2 to 99.5).
Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: an observational study using national surveillance data
[Pfizer.] During the analysis period (Jan 24 to April 3, 2021), there were 232 268 SARS-CoV-2 infections, 7694 COVID-19 hospitalisations, 4481 severe or critical COVID-19 hospitalisations, and 1113 COVID-19 deaths in people aged 16 years or older. By April 3, 2021, 4 714 932 (72·1%) of 6 538 911 people aged 16 years and older were fully vaccinated with two doses of BNT162b2. Adjusted estimates of vaccine effectiveness at 7 days or longer after the second dose were 95·3% (95% CI 94·9–95·7; incidence rate 91·5 per 100 000 person-days in unvaccinated vs 3·1 per 100 000 person-days in fully vaccinated individuals) against SARS-CoV-2 infection. 8006 of 8472 samples tested showed a spike gene target failure, giving an estimated prevalence of the B.1.1.7 variant of 94·5% among SARS-CoV-2 infections.
Vaccine Breakthrough Infections with SARS-CoV-2 Variants
In a cohort of 417 persons who had received the second dose of BNT162b2 (Pfizer–BioNTech) or mRNA-1273 (Moderna) vaccine at least 2 weeks previously, we identified 2 women with vaccine breakthrough infection. Despite evidence of vaccine efficacy in both women, symptoms of Covid-19 developed, and they tested positive for SARS-CoV-2 by PCR. Viral sequencing revealed variants of likely clinical importance, including E484K in 1 woman and three mutations (T95I, del142–144, and D614G) in both. These observations indicate a potential risk of illness after successful vaccination and subsequent infection with variant virus, and they provide support for continued efforts to prevent and diagnose infection and to characterize variants in vaccinated persons.
Efficacy of the ChAdOx1 nCoV-19 Covid-19 Vaccine against the B.1.351 Variant
[AstraZeneca. Reposted from Mar 24.] Between June 24 and November 9, 2020, we enrolled 2026 HIV-negative adults (median age, 30 years); 1010 and 1011 participants received at least one dose of placebo or vaccine, respectively. Both the pseudovirus and the live-virus neutralization assays showed greater resistance to the B.1.351 variant in serum samples obtained from vaccine recipients than in samples from placebo recipients. In the primary end-point analysis, mild-to-moderate Covid-19 developed in 23 of 717 placebo recipients (3.2%) and in 19 of 750 vaccine recipients (2.5%), for an efficacy of 21.9% (95% confidence interval [CI], −49.9 to 59.8). Among the 42 participants with Covid-19, 39 cases (92.9%) were caused by the B.1.351 variant; vaccine efficacy against this variant, analyzed as a secondary end point, was 10.4% (95% CI, −76.8 to 54.8). A two-dose regimen of the ChAdOx1 nCoV-19 vaccine did not show protection against mild-to-moderate Covid-19 due to the B.1.351 variant.
Preliminary Analysis of Safety and Immunogenicity of a SARS-CoV-2 Variant Vaccine Booster
[Moderna booster. Preprint.] Here we present the preliminary evaluation of a clinical study on the use of the prototype mRNA-1273 or modified COVID-19 mRNA vaccines, designed to target emerging SARS-CoV-2 variants as booster vaccines in participants previously vaccinated approximately 6 months earlier with two doses of the prototype vaccine, mRNA-1273. The modified vaccines include a monovalent mRNA-1273.351 encoding for the S protein found in the B.1.351 variant and multivalent mRNA-1273.211 comprising a 1:1 mix of mRNA-1273 and mRNA-1273.351. As single 50 µg booster vaccinations, both mRNA-1273 and mRNA-1273.351 had acceptable safety profiles and were immunogenic. Antibody neutralization titers against B.1.351 and P.1 variants measured by SARS-CoV-2 pseudovirus neutralization (PsVN) assays before the booster vaccinations, approximately 6 to 8 months after the primary series, were low or below the assay limit of quantification, although geometric mean titers versus the wild-type strain remained above levels likely to be protective. Two weeks after the booster vaccinations, titers against the wild-type original strain, B.1.351, and P.1 variants increased to levels similar to or higher than peak titers after the primary series vaccinations. Although both mRNA-1273 and mRNA-1273.351 boosted neutralization of the wild-type original strain, and B.1.351 and P.1 variants, mRNA-1273.351 appeared to be more effective at increasing neutralization of the B.1.351 virus versus a boost with mRNA-1273. The vaccine trial is ongoing.
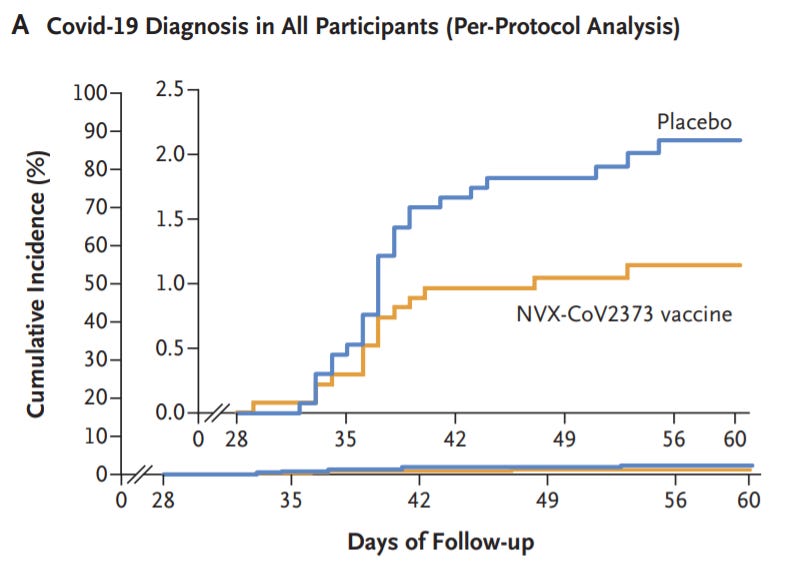
Efficacy of NVX-CoV2373 Covid-19 Vaccine against the B.1.351 Variant
[Novavax booster.] In this phase 2a–b trial in South Africa […] 6324 participants underwent screening and 4387 received at least one injection of vaccine or placebo. Approximately 30% of the participants were seropositive for SARS-CoV-2 at baseline. Among 2684 baseline seronegative participants (94% HIV-negative and 6% HIV-positive), predominantly mild-to-moderate Covid-19 developed in 15 participants in the vaccine group and in 29 in the placebo group (vaccine efficacy, 49.4%; 95% confidence interval [CI], 6.1 to 72.8). Vaccine efficacy among HIV-negative participants was 60.1% (95% CI, 19.9 to 80.1). Of 41 sequenced isolates, 38 (92.7%) were the B.1.351 variant. Post hoc vaccine efficacy against B.1.351 was 51.0% (95% CI, −0.6 to 76.2) among the HIV-negative participants.