Effectiveness of vaccines against Delta variant
Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant
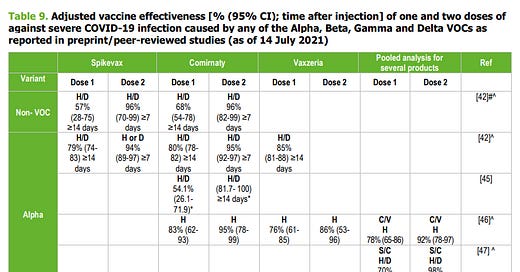
Data on all symptomatic sequenced cases of Covid-19 in England were used to estimate the proportion of cases with either variant according to the patients’ vaccination status. Effectiveness after one dose of vaccine (BNT162b2 or ChAdOx1 nCoV-19) was notably lower among persons with the delta variant (30.7%; 95% confidence interval [CI], 25.2 to 35.7) than among those with the alpha variant (48.7%; 95% CI, 45.5 to 51.7); the results were similar for both vaccines. With the BNT162b2 vaccine, the effectiveness of two doses was 93.7% (95% CI, 91.6 to 95.3) among persons with the alpha variant and 88.0% (95% CI, 85.3 to 90.1) among those with the delta variant. With the ChAdOx1 nCoV-19 vaccine, the effectiveness of two doses was 74.5% (95% CI, 68.4 to 79.4) among persons with the alpha variant and 67.0% (95% CI, 61.3 to 71.8) among those with the delta variant. Only modest differences in vaccine effectiveness were noted with the delta variant as compared with the alpha variant after the receipt of two vaccine doses.
Myocarditis with COVID-19 mRNA Vaccines
According to the U.S. Centers for Disease Control (CDC), myocarditis/pericarditis rates are approximately 12.6 cases per million doses of second dose mRNA vaccine among 12-39-year-olds. In reported cases, patients with myocarditis invariably presented with chest pain, usually 2-3 days after a second dose of mRNA vaccination and had elevated cardiac troponin levels. Almost all patients had resolution of symptoms and signs, and improvement in diagnostic markers and imaging with or without treatment. Despite rare cases of myocarditis, the benefit-risk assessment for COVID-19 vaccination shows a favorable balance for all age and sex groups.
Neutralizing activity of Sputnik V vaccine sera against SARS-CoV-2 variants
Here, we characterize the neutralizing activity of post-Sputnik V vaccination sera against the ensemble of S mutations present in alpha (B.1.1.7) and beta (B.1.351) variants of concern (VOC). We determine that only 1 out of 12 post-vaccination serum samples shows effective neutralization (IC90) of rcVSV-CoV2-S: B.1.351 at full serum strength. The same set of sera efficiently neutralize S from B.1.1.7 and exhibit only moderately reduced activity against S carrying the E484K substitution alone. Taken together, our data suggest that control of some emergent SARS-CoV-2 variants may benefit from updated vaccines.
US COVID-19 hospital cases surge
[Media report.] After an early summer drop, cases are rising across the country as the Delta variant (B1617.2) spreads and under-vaccinated communities remain susceptible to the virus. Similarly, almost every state is seeing an increase in COVID-19 hospitalizations, especially Nevada, Arkansas, and Missouri. The only states not seeing hospitalizations grow are Maryland, North Dakota, Pennsylvania, Rhode Island, and Vermont. [See also Global COVID deaths rise sharply.]
Partial COVID-19 vaccination, vaccination following SARS-CoV-2 infection and heterologous vaccination schedule: summary of evidence
[ECDC technical report.] Preliminary evidence from some studies indicates that individuals who are partially vaccinated are less protected against symptomatic infection with the B.1.617.2 (Delta) VOC than against the B.1.1.7 (Alpha) VOC, regardless of the vaccine type. However, full vaccination provides nearly equivalent protection against the Delta to that for the Alpha VOC. Studies of single-dose regimens of Comirnaty, Spikevax and Vaxzevria in previously infected individuals indicate antibody and cellular immune responses are comparable to naïve individuals who complete the two-dose regimen. However, data on the long-term duration of protective immunity are sparse. Given the current evidence gaps, as a precaution, consideration should be given to the continued administration of a two-dose regimen, as per EMA authorisation, particularly for those individuals at greatest risk of severe outcomes following SARS-CoV-2 infection. Evidence from studies on heterologous (‘mix and match’) vaccination suggests that the combination of Vaxzevria and mRNA vaccines induces a robust humoral response against SARS-CoV-2 and elicits a higher T-cell response than homologous combinations. Although increased mild-to-moderate systemic reactogenicity was observed after administration of the second dose, heterologous regimens were generally well tolerated.