Emergence and spread of new variant in Mexico
Emergence and spread of the potential variant of interest (VOI) B.1.1.519 predominantly present in Mexico
[Preprint.] In the present work, we are reporting the identification of a potential variant of interest harboring the mutations T478K, P681H, and T732A in the Spike protein, within the newly named lineage B.1.1.519, which rapidly outcompeted the preexisting variants in Mexico and has been the dominant virus in the country during the first trimester of 2021.
Effectiveness of the CoronaVac vaccine in the elderly population during a P.1 variant-associated epidemic of COVID-19 in Brazil: A test-negative case-control study
[Preprint.] We conducted a test-negative, case-control study of adults ≥70 years of age from São Paulo State from January 17 to April 29, 2021, during which vaccination with a two-dose regimen of CoronaVac was implemented. We selected 7,950 matched pairs with a mean age of 76 years from 26,433 COVID-19 cases and 17,622 test-negative controls. Adjusted vaccine effectiveness was 18.2% (95% CI, 0.0 to 33.2) and 41.6% (95% CI, 26.9 to 53.3) in the period 0-13 and ≥14 days, respectively, after the 2nd dose. Administration of a single vaccine dose was not associated with reduced odds of COVID-19.
Interim Estimates of Vaccine Effectiveness of Pfizer-BioNTech and Moderna COVID-19 Vaccines Among Health Care Personnel - 33 U.S. Sites, January-March 2021
A test-negative case-control study is underway to evaluate mRNA COVID-19 vaccine effectiveness (VE) against symptomatic illness among HCP at 33 U.S. sites across 25 U.S. states. Interim analyses indicated that the VE of a single dose (measured 14 days after the first dose through 6 days after the second dose) was 82% (95% confidence interval [CI] = 74%-87%), adjusted for age, race/ethnicity, and underlying medical conditions. The adjusted VE of 2 doses (measured ≥7 days after the second dose) was 94% (95% CI = 87%-97%). VE of partial (1-dose) and complete (2-dose) vaccination in this population is comparable to that reported from clinical trials and recent observational studies, supporting the effectiveness of mRNA COVID-19 vaccines against symptomatic disease in adults, with strong 2-dose protection.
BNT162b2 mRNA Covid-19 vaccine effectiveness in the prevention of SARS-CoV-2 infection: a preliminary report
[Pfizer.] Bari Policlinico University-Hospital healthcare workers (HCWs) who completed the vaccination schedule were matched with HCWs who had refused vaccination. The VE for documented infection was 61.9% (95%CI=19.2–82.0%) 14–20 days after the first dose, 87.9% (95%CI=51.7–97.0%) 21–27 days after the first dose, and 96.0% (95%CI=82.2–99.1) 7 or more days after the second dose. Unvaccinated HCWs remain a concern even in the context of pandemic emergency.
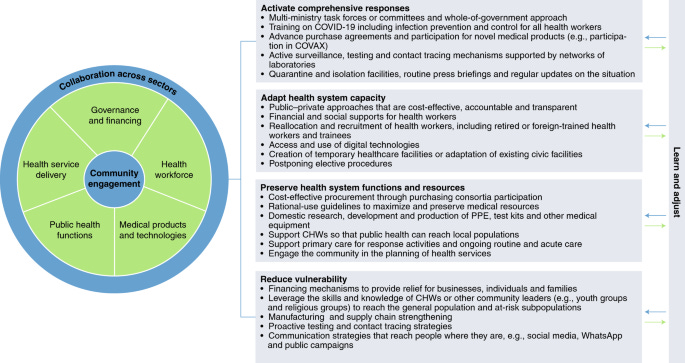
Health systems resilience in managing the COVID-19 pandemic: lessons from 28 countries
We review COVID-19 responses in 28 countries using a new health systems resilience framework. We report on domains addressing governance and financing, health workforce, medical products and technologies, public health functions, health service delivery and community engagement to prevent and mitigate the spread of COVID-19.High-performing countries activated comprehensive responses across the determinants’ domains, including through whole-of-government approaches and the creation of multi-ministry task forces, to ensure adequate translation of evidence into policy and practices that preserve health system capacity, while protecting public health and livelihoods. Specific measures taken include training health workers, bolstering public health functions (including offering designated isolation facilities, either for all or for those unable to safely self-isolate at home) and preparing for new technologies and medicines through purchase agreements, while also engaging communities through routine communications on the epidemiological situation and emergent policies.