Inactivated vaccine 59% effective vs Delta infection, 70% vs moderate Covid
Efficacy of inactivated SARS-CoV-2 vaccines against the Delta variant infection in Guangzhou: A test-negative case-control real-world study
We recruited participants aged 18-59 years who consisted of SARS-CoV-2 test-positive cases (n=74) and test-negative controls (n=292) during the outbreak of the Delta variant in May 2021 in Guangzhou city, China. A single dose of inactivated SARS-CoV-2 vaccine yielded the VE of only 13.8%. After adjusting for age and sex, the overall VE for two-dose vaccination was 59.0% (95% confidence interval: 16.0% to 81.6%) against coronavirus disease 2019 (COVID-19) and 70.2% (95% confidence interval: 29.6% to 89.3%) against moderate COVID-19 and 100% against severe COVID-19 which might overestimate due to the small sample size.
Low infectious risk of re-positive COVID-19 patients: a single-center study
A total 255 (16.04%) out of 1590 COVID-19 cases were re-positive. The re-positives were more likely to occur in patients at the age of 20-39 and patients with moderate-severity. Quantitative PCR showed CT value and viral loads were both far lower than the hospitalized COVID-19 patients. The viral culture of the sample from re-positive cases showed no cytopathic effect, and NAT for the culture medium of viral culture all exhibited negative results. The viral load of the re-positive cases was very low, not infectious, and the risk of human-to-human transmission was extremely low. The discharged COVID-19 cases should undergo home health management for 3 weeks.
A systematic review of the impact of the Alpha and Gamma variants of concern on hospitalization and symptomatic rates of SARS-CoV-2
[Preprint.] The firmest conclusion of this review is that the Alpha VOC comes with a higher relative risk of hospitalization compared to the original wildtype, most likely above 1.4, while unlikely to be above 2, with the balance of evidence being that the relative risk is not significantly modified by age. The evidence for Gamma is more limited, but the odds ratio may be above 2, and potentially much greater than that, especially for those 20-39 years of age.
Effect of anakinra on mortality in patients with COVID-19: a systematic review and patient-level meta-analysis
209 articles were identified, of which 178 full-text articles fulfilled screening criteria and were assessed. Aggregate data on 1185 patients from nine studies were analysed, and individual patient-level data on 895 patients were provided from six of these studies. Eight studies were observational and one was a randomised controlled trial. In the individual patient-level meta-analysis, after adjusting for age, comorbidities, baseline ratio of the arterial partial oxygen pressure divided by the fraction of inspired oxygen (PaO2/FiO2), C-reactive protein (CRP) concentrations, and lymphopenia, mortality was significantly lower in patients treated with anakinra (38 [11%] of 342) than in those receiving standard of care with or without placebo (137 [25%] of 553; adjusted odds ratio [OR] 0·32 [95% CI 0·20–0·51]). Anakinra was not associated with a significantly increased risk of secondary infections when compared with standard of care (OR 1·35 [95% CI 0·59–3·10]). Anakinra could be a safe, anti-inflammatory treatment option to reduce the mortality risk in patients admitted to hospital with moderate to severe COVID-19 pneumonia.
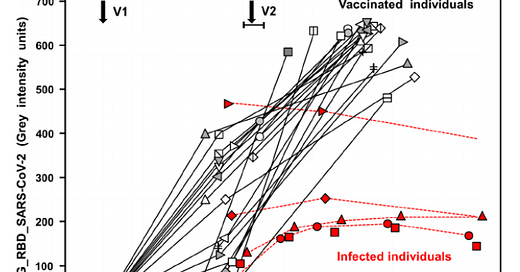
Vaccination versus infection with SARS-CoV-2: Establishment of a high avidity IgG response versus incomplete avidity maturation
We confirm and extend data on incomplete avidity maturation of IgG toward severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) nucleoprotein (NP), spike protein-1 (S1), and its receptor-binding domain (RBD) in coronavirus disease 2019 (COVID-19) patients. In SARS-CoV-2-infected individuals, an initial rise in avidity maturation was ending abruptly, leading to IgG of persistently low or intermediate avidity. Incomplete avidity maturation might facilitate secondary SARS-CoV-2 infections and thus prevent the establishment of herd immunity. Incomplete avidity maturation after infection with SARS-CoV-2 (with only 11.8% of cases showing finally IgG of high avidity, that is, an avidity index > 0.6) was contrasted by regular and rapid establishment of high avidity in SARS-CoV-2 naïve individuals after two vaccination steps with the BioNTech messenger RNA (mRNA) Vaccine (78% of cases with high avidity). One vaccination step was not sufficient for induction of complete avidity maturation in vaccinated SARS-CoV-2 naïve individuals, as it induced high avidity only in 2.9% of cases within 3 weeks. However, one vaccination step was sufficient to induce high avidity in individuals with previous SARS-CoV-2 infection.