Indian variant escapes antibodies partially
Reduced sensitivity of infectious SARS-CoV-2 variant B.1.617.2 to monoclonal antibodies and sera from convalescent and vaccinated individuals
[Preprint.] We isolated infectious B.1.617.2 from a traveller returning from India. We examined its sensitivity to monoclonal antibodies (mAbs) and to antibodies present in sera from COVID-19 convalescent individuals or vaccine recipients. B.1.617.2 was resistant to neutralization by some anti-NTD and anti-RBD mAbs, including Bamlanivimab, which were impaired in binding to the B.1.617.2 Spike. Sera from convalescent patients collected up to 12 months post symptoms and from Pfizer Comirnaty vaccine recipients were 3 to 6 fold less potent against B.1.617.2, relative to B.1.1.7. Sera from individuals having received one dose of AstraZeneca Vaxzevria barely inhibited B.1.617.2. Thus, B.1.617.2 spread is associated with an escape to antibodies targeting non-RBD and RBD Spike epitopes.
Infection and vaccine-induced neutralizing antibody responses to the SARS-CoV-2 B.1.617.1 variant
[Preprint.] In this study, using a live virus assay, we describe the neutralizing antibody response to the B.1.617.1 variant in serum from infected and vaccinated individuals. We found that the B.1.617.1 variant is 6.8-fold more resistant to neutralization by sera from COVID-19 convalescent and Moderna and Pfizer vaccinated individuals. Despite this, a majority of the sera from convalescent individuals and all sera from vaccinated individuals were still able to neutralize the B.1.617.1 variant. This suggests that protective immunity by the mRNA vaccines tested here are likely retained against the B.1.617.1 variant.
Acute Coronavirus Disease (COVID-19) in Vaccinated Frontline Healthcare Workers in India: An Observational Cohort Study
[Preprint.] Healthcare workers (HCWs) in India who developed acute Covid-19 infection despite receiving at least 1 dose of the vaccination were invited to complete an online questionnaire. Out of contacted 2160 HCWs, 316 (14.6%) developed acute Covid-19 infection post- vaccination. Of the 316 participants, 50 (15.8%) had developed Covid-19 after receiving only the first dose of the vaccine. Participants who completed both doses of vaccine (n=266) were more likely to be asymptomatic (13.9% vs 2.0%, p=0.016), but clinical outcomes such as hospitalization, need for mechanical ventilation and supplemental oxygen were similar. Participants with pneumonia (n=105, 33.2%) had elevated C-reactive protein (CRP, 25.4±21.2 vs 6.8±8.8 mg/L, p<0.001) compared with those without, and were more likely to require hospitalization (30.5% vs 6.2%, p<0.001) and supplemental oxygen (9.5% vs 0.0%, p<0.001). On multivariable analyses, the completion of both doses of vaccination was protective for the development of fever in Covid-19 illness (adjusted OR 0.33, 95%CI 0.15-0.72), after adjusting for age, prior medical conditions and CRP. Post-vaccination antibody titres to the spike protein were not significantly associated with symptoms or outcomes.
Characterization of the emerging B.1.621 variant of interest of SARS-CoV-2
[Preprint.] Here, we report the emergence of the B.1.621 lineage, considered a variant of interest (VOI) with the accumulation of several mutations affecting the Spike protein, including the amino acid changes I95I, Y144T, Y145S and the insertion 146N in the N-terminal domain, R346K, E484K and N501Y in the Receptor Binding Domain and P681H in the S1/S2 cleavage site of the Spike protein. The rapid increase in frequency and fixation in a relatively short time in some cities that were near the theoretical herd immunity suggests an epidemiologic impact.
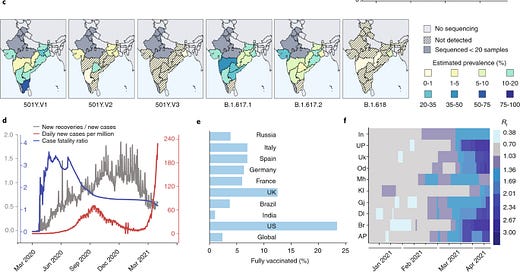
SARS-CoV-2 variants of concern are emerging in India
The sudden surge in COVID-19 cases in India coincides with high prevalence of more-transmissible variants, associated with diagnostic test failures and antibody escape. Confirmed daily new cases in India have spiked from 53 per million population (up to March 2021) to >200 per million population (after mid-March 2021) (Fig. 1d). A glimmer of hope lies in an apparently low case-fatality ratio of <1.5%, compared with >3% between July 2020 and August 2020 (Fig. 1d), although many researchers question the official fatality rates. New variants are thought to be responsible for re-infections, either after natural infection or after vaccination, as observed in Brazil and the United States, respectively. There is evidence that re-infections are already happening in India; a recent survey identified a re-infection proportion of 4.5% from a pool of 1,300 participants infected between January 2020 and October 2020.