Molnupiravir, an oral antiviral treatment for COVID-19
Covid-19: Molnupiravir reduces risk of hospital admission or death by 50% in patients at risk, Merck reports
The antiviral drug molnupiravir reduced the risk of admission to hospital or death by around 50% in non-hospitalised adults who had mild to moderate covid-19 and were at risk of poor outcomes, Merck Sharp and Dohme (MSD) has reported. The company said in a press release that 7.3% of patients (28 of 385) who received molnupiravir as part of the phase III trial and 14.1% of patients taking placebo (53 of 377) either were admitted to hospital or had died by day 29 after randomisation. [See also Merck press release.]
Molnupiravir, an Oral Antiviral Treatment for COVID-19
[Preprint.] Eligible participants included outpatients with confirmed SARS-CoV-2 infection and symptom onset within 7 days. Among 202 treated participants, virus isolation was significantly lower in participants receiving 800 mg molnupiravir (1.9%) versus placebo (16.7%) at Day 3 (p = 0.02). At Day 5, virus was not isolated from any participants receiving 400 or 800 mg molnupiravir, versus 11.1% of those receiving placebo (p = 0.03). Time to viral RNA clearance was decreased and a greater proportion overall achieved clearance in participants administered 800 mg molnupiravir versus placebo (p = 0.01). Molnupiravir was generally well tolerated, with similar numbers of adverse events across all groups.
Human Safety, Tolerability, and Pharmacokinetics of Molnupiravir, a Novel Broad-Spectrum Oral Antiviral Agent with Activity against SARS-CoV-2
Single and multiple doses of molnupiravir were evaluated in this first-in-human, phase 1, randomized, double-blind, placebo-controlled study in healthy volunteers, which included evaluation of the effect of food on pharmacokinetics. Molnupiravir (EIDD-1931) appeared rapidly in plasma, with a median time of maximum observed concentration of 1.00 to 1.75 h, and declined with a geometric half-life of approximately 1 h, with a slower elimination phase apparent following multiple doses or higher single doses (7.1 h at the highest dose tested). Mean maximum observed concentration (Cmax) and area under the plasma concentration versus time curve (AUC) increased in a dose-proportional manner, and there was no accumulation following multiple doses. When administered in a fed state, there was a decrease in the rate of absorption, but no decrease in overall exposure. Molnupiravir was well tolerated. Fewer than half of the subjects reported an adverse event, the incidence of adverse events was higher following administration of placebo, and 93.3% of adverse events were mild. One subject discontinued early due to rash. There were no serious adverse events.
Molnupiravir: coding for catastrophe
Molnupiravir, a wide-spectrum antiviral that is currently in phase 2/3 clinical trials for the treatment of COVID-19, is proposed to inhibit viral replication by a mechanism known as ‘lethal mutagenesis’. Two recently published studies reveal the biochemical and structural bases of how molnupiravir disrupts the fidelity of SARS-CoV-2 genome replication and prevents viral propagation by fostering error accumulation in a process referred to as ‘error catastrophe’.
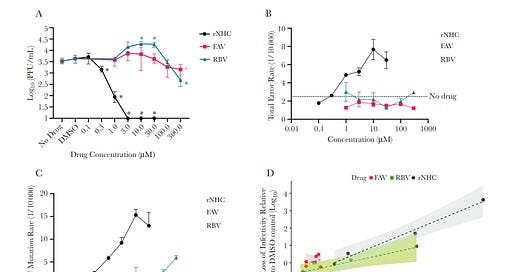
β-D-N4-hydroxycytidine Inhibits SARS-CoV-2 Through Lethal Mutagenesis But Is Also Mutagenic To Mammalian Cells
β-D-N4-hydroxycytidine (NHC, initial metabolite of molnupiravir) is >100-fold more active than ribavirin or favipiravir against SARS-CoV-2, with antiviral activity correlated to the level of mutagenesis in virion RNA. However, NHC also displays host mutational activity in an animal cell culture assay, consistent with RNA and DNA precursors sharing a common intermediate of a ribonucleoside diphosphate. These results indicate highly active mutagenic ribonucleosides may hold risk for the host.
Related news headlines: