Prior infection protects against reinfection, even in the absence of antibodies
Prior COVID-19 protects against reinfection, even in the absence of detectable antibodies
We examined datasets from four UK laboratories and identified a subset of patients with proven SARS-CoV-2 infection, defined as laboratory detection of RNA, in the first wave of the pandemic between March and May 2020, but with negative serology results in June and July. We identified 224 RNA-positive antibody-negative patients in the first wave, with two laboratory-confirmed reinfections in the second wave (0.89%), compared to 2054 second-wave infections in the 47139 patients with previous negative serology and either no RNA result, or a negative RNA result (4.36%.) This implies a significantly reduced risk of reinfection (relative risk 0.20, 95% CI 0.05 to 0.81) in those with prior SARS-CoV-2 infection but without detectable antibodies, compared to those with no previous evidence of infection. We also found 2087 RNA-positive antibody positive patients in the first wave, with 18 reinfections (0.86%) – this was similar to the proportion in the RNA-positive antibody-negative patients (relative risk 1.04). Our results indicate that antibodies (as detected by routine laboratory assays) are not essential for protection against reinfection.
Mortality Rate and Characteristics of Deaths Following COVID-19 Vaccination
Data on deaths following COVID-19 vaccination were obtained from the Vaccine Adverse Event Reporting System (VAERS) from December 11, 2020 through January 8, 2021. As of January 8, 2021, 55 deaths were reported, and the mortality rate of COVID-19 vaccination was 8.2 per million population. A total of 37 deaths were reported among long-term care facility residents, and the mortality rate was 53.4 per million population. Top reported comorbidities associated with deaths included hypertension, dementia, chronic obstructive pulmonary disease (COPD), diabetes, and heart failure. In addition, dementia was more likely to be associated with deaths vaccinated at long-term care facilities than at other locations. The benefits of COVID-19 vaccines outweigh the potential risks in older frail populations.
Novavax COVID-19 Vaccine Demonstrates 90% Overall Efficacy and 100% Protection Against Moderate and Severe Disease in PREVENT-19 Phase 3 Trial
[Press release.] Novavax […] today announced that NVX-CoV2373, its recombinant nanoparticle protein-based COVID-19 vaccine, demonstrated 100% protection against moderate and severe disease, 90.4% efficacy overall, and met the primary endpoint in its PREVENT-19 pivotal Phase 3 trial. The study enrolled 29,960 participants across 119 sites in the U.S. and Mexico to evaluate efficacy, safety and immunogenicity, with an emphasis on recruiting a representative population of communities and demographic groups most impacted by the disease.
A scoping review of research on the determinants of adherence to social distancing measures during the COVID-19 pandemic
The review included 84 studies. Many lacked theoretical input, were at risk for bias, and few were experimental in design. The most commonly coded domains of the TDF in the included studies were ‘Environmental Context and Resources’ (388 codes across 76 studies), ‘Beliefs about Consequences’ (34 codes across 21 studies), ‘Emotion’ (28 codes across 12 studies), and ‘Social Influences’ (26 codes across 16 studies). The least frequently coded TDF domains included ‘Optimism’ (not coded), ‘Intentions’ (coded once), ‘Goals’ (2 codes across 2 studies), ‘Reinforcement’ (3 codes across 2 studies), and ‘Behavioural Regulation’ (3 codes across 3 studies). The quality of the included studies was variable and their generalisablity was threatened by their reliance on convenience samples.
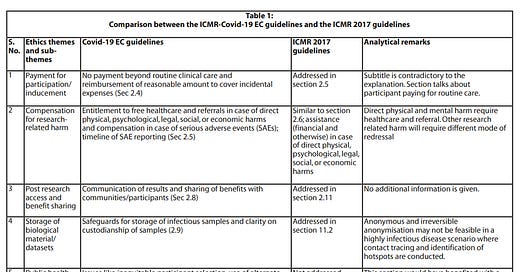
National Guidelines for Ethics Committees Reviewing Biomedical & Health Research during COVID-19 Pandemic: An analysis
The Indian Council of Medical Research has developed the “National Guidelines for Ethics Committees Reviewing Biomedical & Health Research during Covid-19 Pandemic” for guidance during the review of research. Here, we attempt to analyse the strengths and limitations of these guidelines to assess if the unique ethical challenges faced during research in the current situation are adequately identified and addressed and if foundational values and principles of research ethics have been taken into account in these guidelines.