Reinfection with variants (breakthrough infections)
Case series of four re-infections with a SARS-CoV-2 B.1.351 variant, Luxembourg, February 2021
We describe a series of four cases of re-infection with the B1.351 variant in Luxembourg, in 2021, in healthcare workers who had been previously been infected with SARS-CoV-2 in 2020, before the B1.351 variant was first detected in Europe. To our knowledge, only one other case of re-infection with the B1.351 variant has been described in the literature thus far. The patient had severe illness requiring intensive care and mechanical ventilation. The four HCWs reported here were all young without comorbidities, they all recovered and their re-infections were not severe. Moreover, the re-infection was not more severe than the first episode for each of them.
Variants of concern are overrepresented among post-vaccination breakthrough infections of SARS-CoV-2 in Washington State
[Preprint.] Across 20 vaccine breakthrough cases detected at our institution, all 20 (100%) infections were due to variants of concern (VOC) and had a median Ct of 20.2 (IQR=17.1-23.3). When compared to 5174 contemporaneous samples sequenced in our laboratory, VOC were significantly enriched among breakthrough infections (p < .05).
Antigenic minimalism of SARS-CoV-2 is linked to surges in COVID-19 community transmission and vaccine breakthrough infections
[Preprint.] Here, we systematically analyzed over 1.3 million SARS-CoV-2 genomes from 178 countries and performed whole-genome viral sequencing from 53 COVID-19 patients, including 20 vaccine breakthrough infections. We identified 116 Spike protein mutations. Deletions in the Spike protein N-terminal domain (NTD) are highly enriched for these ‘surge-associated mutations’ (Odds Ratio = 18.2, 95% CI: 7.53-48.7; p=1.465×10−18). Overall, the expanding repertoire of Spike protein N-terminal domain deletions throughout the pandemic and their association with case surges and vaccine breakthrough infections point to antigenic minimalism as an emerging evolutionary strategy for SARS-CoV-2 to evade immune responses.
SARS-CoV-2 variants of concern partially escape humoral but not T-cell responses in COVID-19 convalescent donors and vaccinees
Here, we studied humoral and cellular immune responses to wild type SARS-CoV-2 and the B.1.1.7 and B.1.351 variants of concern in a cohort of 121 BNT162b2 mRNA-vaccinated health care workers (HCW). Twenty-three HCW recovered from mild COVID-19 disease and exhibited a recall response with high levels of SARS-CoV-2-specific functional antibodies and virus-specific T cells after a single vaccination. Vaccination-induced antibodies cross-neutralized the variants B.1.1.7 and B.1.351, but the neutralizing capacity and Fc-mediated functionality against B.1.351 was consistently 2- to 4-fold lower than to the homologous virus. Importantly, we observed no differences in CD4+ T-cell activation in response to variant antigens, indicating that the B.1.1.7 and B.1.351 S proteins do not escape T-cell-mediated immunity elicited by the wild type S protein. In conclusion, this study shows that some variants can partially escape humoral immunity induced by SARS-CoV-2 infection or BNT162b2 vaccination, but S-specific CD4+ T-cell activation is not affected by the mutations in the B.1.1.7 and B.1.351 variants.
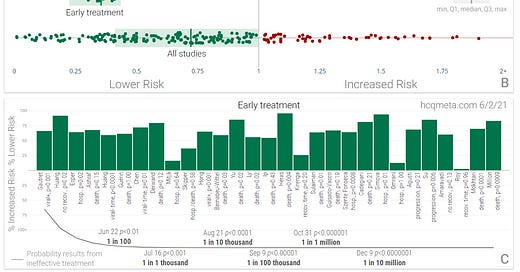
Vitamin D for COVID-19: real-time meta analysis of 80 studies
[Open source.] We analyze all significant studies regarding vitamin D and COVID-19. 96% of 25 vitamin D treatment studies report positive effects (14 statistically significant in isolation). Random effects meta-analysis with pooled effects using the most serious outcome reported shows 78% and 56% improvement for early treatment and for all studies (RR 0.22 [0.12-0.39] and 0.44 [0.34-0.58]). Results are similar after restriction to 20 peer-reviewed studies: 83% and 58% (RR 0.17 [0.07-0.42] and 0.42 [0.31-0.57]), and for the 15 mortality results: 78% and 65% (RR 0.22 [0.12-0.43] and 0.35 [0.24-0.52]). Sufficiency studies show a strong association between vitamin D sufficiency and outcomes. Meta analysis of the 55 studies with pooled effects using the most serious outcome reported shows 51% improvement (RR 0.49 [0.42-0.57]). Heterogeneity arises from many factors including treatment delay, patient population, the effect measured, variants, the form of vitamin D used, and treatment regimens. While many treatments have some level of efficacy, they do not replace vaccines and other measures to avoid infection.
Hydroxychloroquine for COVID-19: real-time meta analysis of 248 studies
[Open source.] 298 studies, 222 peer reviewed, 248 comparing treatment and control groups. Random effects meta-analysis with pooled effects using the most serious outcome reported shows 66% improvement for the 29 early treatment studies (RR 0.34 [0.24-0.49]). Results are similar after exclusion based sensitivity analysis: 67% (RR 0.33 [0.24-0.44]), and after restriction to 20 peer-reviewed studies: 65% (RR 0.35 [0.25-0.47]). Restricting to the 6 RCTs shows 46% improvement (RR 0.54 [0.33-0.86]). Restricting to the 13 mortality results shows 75% lower mortality (RR 0.25 [0.16-0.40]). Late treatment is less successful, with only 71% of the 168 studies reporting a positive effect. Very late stage treatment is not effective and may be harmful, especially when using excessive dosages. 87% of RCTs for early, Pre-Exposure Prophylaxis, or Post-Exposure Prophylaxis treatment report positive effects. Negative meta analyses of hydroxychloroquine generally choose a subset of trials, focusing on late treatment, especially trials with very late treatment and excessive dosages.