Effectiveness Moderna vaccine vs. Delta, Mu, and other emerging variants
Effectiveness of mRNA-1273 against Delta, Mu, and other emerging variants
[Preprint.] The study included 8,153 cases and their matched controls. Two-dose VE (95% confidence interval) was 86.7% (84.3-88.7%) against Delta infection, 98.4% (96.9-99.1%) against Alpha, 90.4% (73.9-96.5%) against Mu, 96-98% against other identified variants, and 79.9% (76.9-82.5%) against unidentified variants. VE against Delta declined from 94.1% (90.5-96.3%) 14-60 days after vaccination to 80.0% (70.2-86.6%) 151-180 days after vaccination. Waning was less pronounced for non-Delta variants. VE against Delta hospitalization was 97.6% (92.8-99.2%). Two doses of mRNA-1273 were highly effective against all SARS-CoV-2 variants. However, VE against Delta moderately declined with increasing time since vaccination.
Comparison of an inactivated Covid19 vaccine-induced antibody response with concurrent natural Covid19 infection
Eighty vaccinated individuals and 80 Covid-19 patients enrolled to the study. Both groups were matched on age, gender and antibody testing time. The anti-S positivity were determined to be 96.2% and 92.5% in vaccinated and patient groups (p=0.303) while the anti-N positivity was 51.2% and 98.8%, respectively (p=<0,0001). The median values for anti-S and anti-N antibodies were statistically significant between both groups. For the patient group, there was a positive correlation between the age and anti-S1 antibody titers (r=0.333; p=0.003) and there was also a statistically significant increase in anti-N antibody titers in time (r=0.505; p=0.0001). The antibody titers in the vaccinated group were lower than the patients group.
The durability of immunity against reinfection by SARS-CoV-2: a comparative evolutionary study
We obtained antibody optical density data for six human-infecting coronaviruses, extending from 128 days to 28 years after infection between 1984 and 2020. Reinfection by SARS-CoV-2 under endemic conditions would likely occur between 3 months and 5·1 years after peak antibody response, with a median of 16 months. This protection is less than half the duration revealed for the endemic coronaviruses circulating among humans (5–95% quantiles 15 months to 10 years for HCoV-OC43, 31 months to 12 years for HCoV-NL63, and 16 months to 12 years for HCoV-229E). For SARS-CoV, the 5–95% quantiles were 4 months to 6 years. The timeframe for reinfection is fundamental to numerous aspects of public health decision making. As the COVID-19 pandemic continues, reinfection is likely to become increasingly common.
Emergence and spread of SARS-CoV-2 lineage B.1.620 with variant of concern-like mutations and deletions
We here describe a SARS-CoV-2 lineage - designated B.1.620 - discovered in Lithuania and carrying many mutations and deletions in the spike protein shared with widespread variants of concern (VOCs), including E484K, S477N and deletions HV69Δ, Y144Δ, and LLA241/243Δ. As well as documenting the suite of mutations this lineage carries, we also describe its potential to be resistant to neutralising antibodies, accompanying travel histories for a subset of European cases, evidence of local B.1.620 transmission in Europe with a focus on Lithuania, and significance of its prevalence in Central Africa owing to recent genome sequencing efforts there. We make a case for its likely Central African origin using advanced phylogeographic inference methodologies incorporating recorded travel histories of infected travellers.
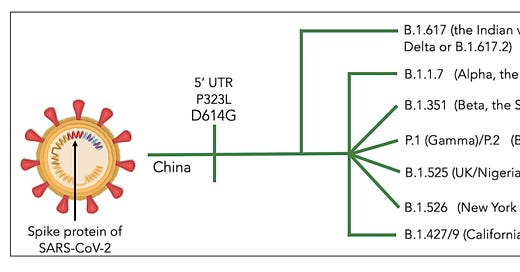
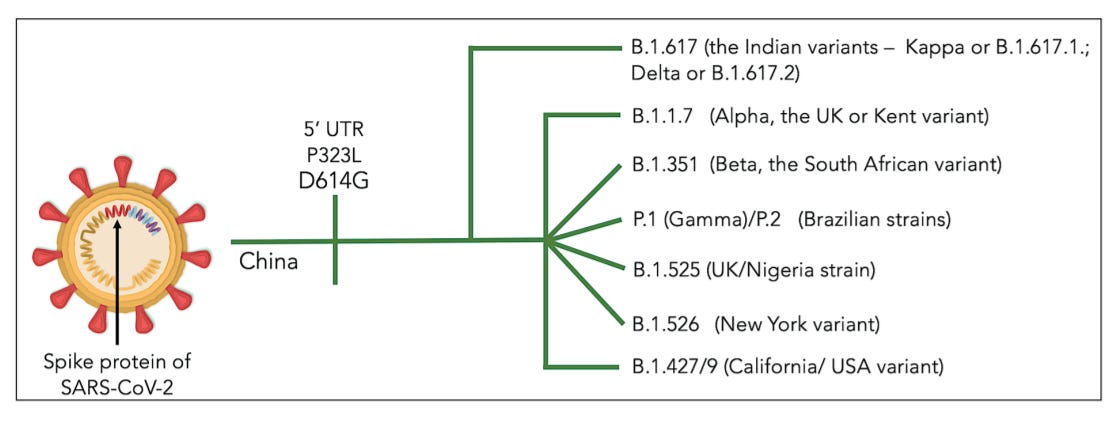
Variants of SARS-CoV-2, their effects on infection, transmission and neutralization by vaccine-induced antibodies
A total of 333 research articles were retrieved through online-database search. Finally, around 35 germane articles were considered for scripting the current report. Mutations have increased the immune escape potential leading to 3.5-6.5-folds decrease in neutralization of antibodies (Pfizer and Moderna vaccines). The variant, B.1.617 circulating in India and many other countries (double variant) having E484Q and L452R mutations, has raised the infection rate and decreased the neutralization capacity of the vaccine-induced antibodies. Deadly K417N+E484K+N501Y triplet mutations found in B.1.351 and P.1 have increased the transmission ability of these strains by 50% leading to greater COVID-19 hospitalization, ICU admissions and deaths. The new SARS-CoV-2 variants have compromised the neutralization potential of the currently used vaccines.